| |
Peter Mayinger
PhD 1992 Ludwig-Maximilians University, Munich, Germany, Postdoctoral work at UCLA, Los Angeles, USA, at ZMBH since 1996, Project Group Leader since 2000
Phosphoinositide Signaling and Transport Processes at Membranes
- Current Research
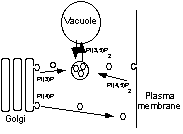
Phosphoinositides are essential phospholipids that serve as key regulators for various cellular processes. The level of these signaling molecules is regulated by specific lipid kinases and phosphatases. We have identified the Sac1 phosphoinositide phosphatase as an important regulator of distinct steps in cellular secretion in yeast. Sac1p seems to have compartment specific roles at the ER and at the Golgi. At the ER Sac1p regulates the uptake of ATP into the ER lumen, which is essential for protein translocation and protein folding in the ER lumen. At the Golgi Sac1p regulates vesicular transport together with the phosphatidylinositol 4-kinase Pik1p. In addition, we study the microsomal ATP-transport system. So far we could define this transport as a specific ADP/ATP-antiport. And we have obtained evidence that the ATP-uptake into the ER is directly regulated by specific phospho-inositides. One main focus of our ongoing research is the search for targets of Sac1p-dependent phosphoinositide signals. One such potential effector protein is the microsomal ATP transporter. Additionally, we want to understand how Sac1p-dependent signals are integrated within other cellular signaling pathways.
Contact:
Dr. Peter Mayinger
Zentrum für Molekulare Biologie
Im Neuenheimer Feld 282
69120 Heidelberg
GermanyTel: -49-6221-546823
Fax:-49-6221-545892
email: mayingerp@sun0.urz.uni-heidelberg.de
http://www.zmbh.uni-heidelberg.de/Mayinger/default.html
- Projects for a Doctoral Thesis
Characterization of components involved in microsomal ATP transport.
Search for effectors of Sac1p-dependent phosphoinositide signals.
Genetic analysis of phosphoinositide signaling pathways
Selected Publications
- Mayinger, P. and Meyer, D. I. 1993. An ATP transporter is required for protein translocation into the yeast endo-plasmic reticulum. EMBO J. 12, 659-666.
Mayinger, P., Bankaitis, V. A. and Meyer, D. I. 1995. Sac1p mediates the ATP transport into yeast endoplasmic reticulum that is required for protein translocation. J. Cell Biol. 131, 1377-1386.
- Kearns, B. G., McGee, T. D., Mayinger, P., Gevilaite, A. Phillips, S. E., Kagiwada, S. and Bankaitis, V. A. 1997. An essential role for diacylglycerol in protein transport from the yeast Golgi complex. Nature 387, 101-105.
- Kochendörfer, K.-U., Then, A. R., Kearns, B. G., Bankaitis, V. A., and Mayinger, P. 1999. Sac1p plays a crucial role in microsomal ATP transport, which is distinct from its function in Golgi phospholipid metabolism. EMBO J. 18, 1506-1515.
- Nicolson, T. and Mayinger, P. 2000. Reconstitution of yeast microsomal lipid flip-flop using endogenous aminophospholipids. FEBS Letters 476, 277-281.