Contact us:
Zentrum für Molekulare Biologie der
Universität Heidelberg (ZMBH)
Im Neuenheimer Feld 345
69120 Heidelberg, Germany
Tel.: +49-6221-54 6813
Fax: +49-6221-54 5894
ms-service@zmbh.uni-heidelberg.de
|
Methods & Services Quantitative
proteomics Quantification strategies A 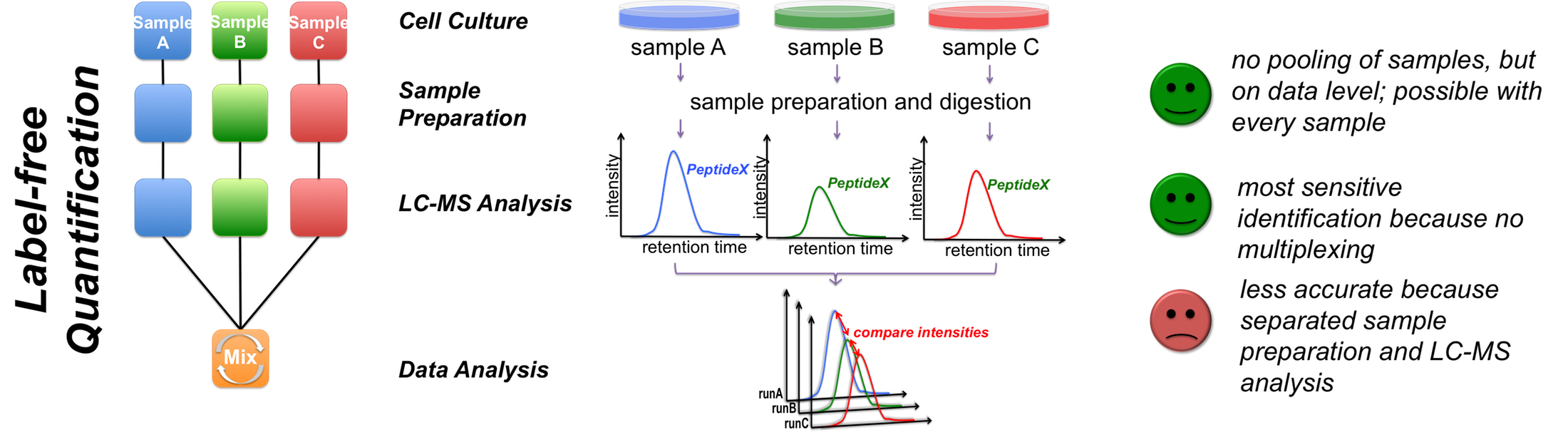
----------------------------------------------------------------------------------------------------------------------------
B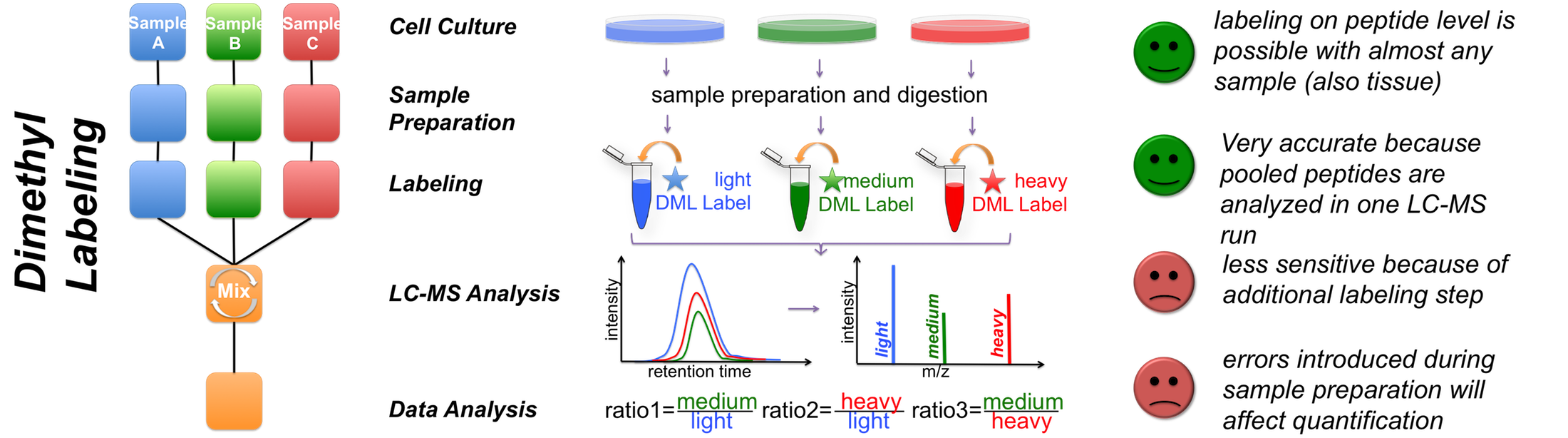
----------------------------------------------------------------------------------------------------------------------------
C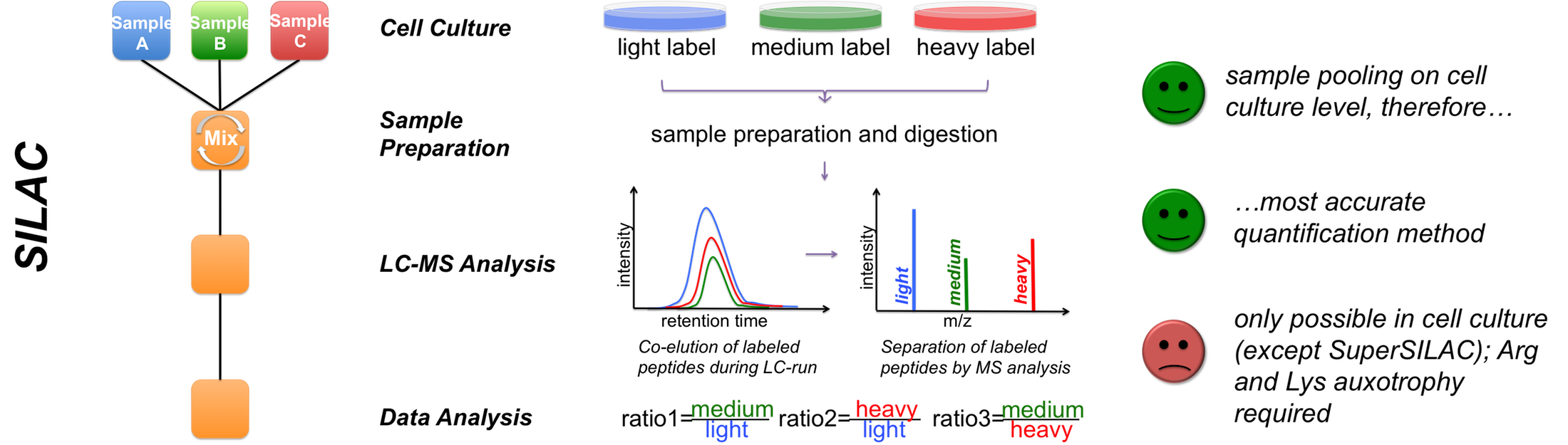 Figure 1 - Quantification strategies available in the Core Facility Targeted proteomics using MRM We also perform targeted analysis, namely MRM (Qtrap 5500), to determine protein complex stoichiometries. The working principle of MRM is shown in Figure 2. 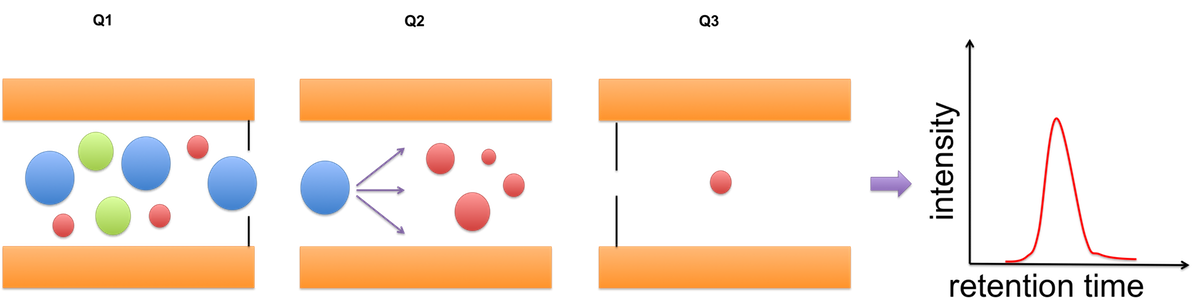 Figure 2 - Principle of an MRM analysis Benchmarking our quantitative MS performance - Succesful participation in ABRF sPRG study 2014 In 2014 we participated in a cross-laboratory study trying to access their quantitative capacities. Therefore a standardized sample containing isotope labeled peptides in a complex HEK293 background was distributed to all participants. As one can observe from the results, our core facility performed very well, especially when using MaxQuant as data analysis software. 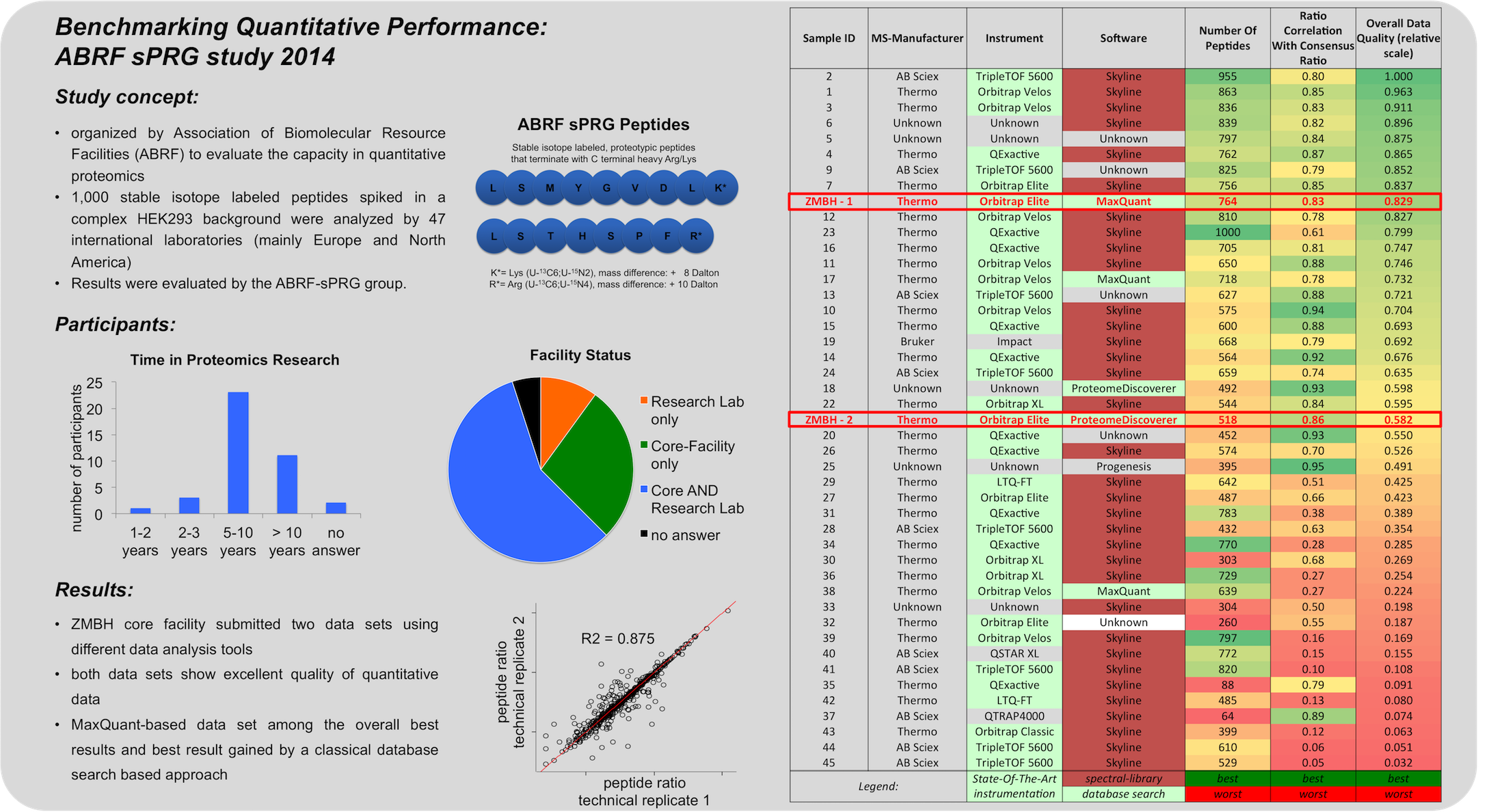 |
|
||